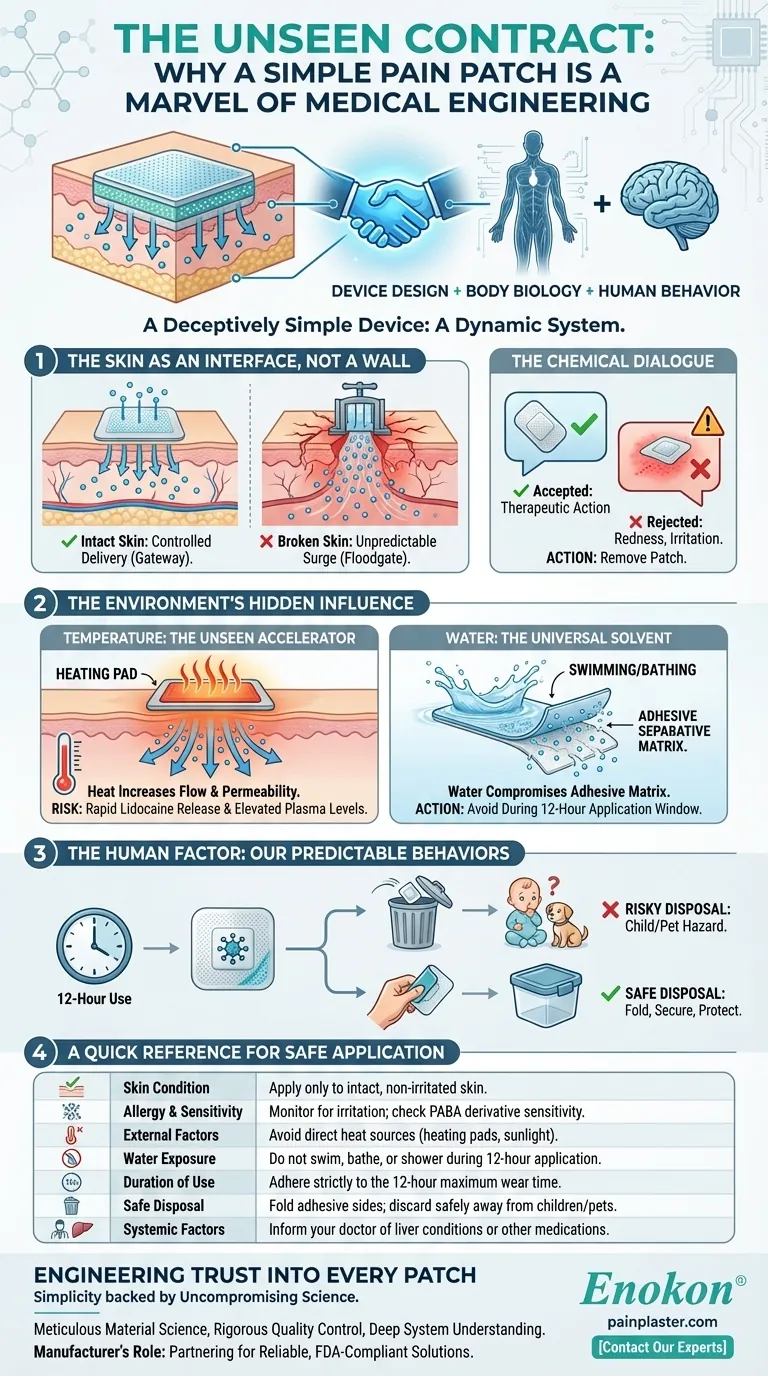
A Deceptively Simple Device
On the surface, a transdermal patch is the picture of simplicity. You peel it, you stick it, and you get localized relief. It feels less like medicine and more like a clever sticker.
This simplicity is a carefully engineered illusion. The patch isn't just a passive object; it's one half of a dynamic system. The other half is you. Its success relies on a silent contract between the device's design, your body's biology, and your behavior. Breaking the terms of this contract, even unintentionally, can undermine its safety and efficacy.
The Skin as an Interface, Not a Wall
We tend to think of our skin as a barrier. But for a transdermal patch, it's an active interface—a gateway for controlled drug delivery. The design of that gateway is paramount.
The Integrity of the System
A lidocaine patch is calibrated to deliver its active ingredient through healthy, intact skin. Applying it to broken, inflamed, or irritated skin is like opening a floodgate. The carefully controlled dose can become an unpredictable surge, increasing systemic absorption and the risk of adverse effects.
The system's integrity relies on this simple rule: the barrier must be respected.
The Chemical Dialogue
The patch also engages in a chemical dialogue with your body. While lidocaine itself is an amide anesthetic, some patients have allergies to a different class—PABA derivatives. While cross-sensitivity isn't guaranteed, it's a known risk.
If your skin signals a problem with redness, irritation, or a burning sensation, it's not a minor inconvenience. It's your body rejecting the terms of the agreement. The patch must be removed.
The Environment's Hidden Influence
A patch doesn't exist in a vacuum. It operates within your daily environment, which contains variables that can fundamentally alter its performance.
Temperature: The Unseen Accelerator
Applying a heating pad over a lidocaine patch seems logical—heat soothes pain. But in this system, it's a dangerous accelerator. Heat increases blood flow and skin permeability, causing the patch to release lidocaine far more rapidly than intended.
This simple act of seeking comfort can elevate plasma lidocaine levels, turning a therapeutic tool into a potential hazard.
Water: The Universal Solvent
The instructions to avoid bathing or swimming with a patch on aren't just about keeping it from falling off. Water can compromise the adhesive matrix that regulates drug release. The 12-hour application window is designed for dry conditions, ensuring the delivery system remains stable and predictable.
The Human Factor: Our Predictable Behaviors
The most unpredictable variable in any system is the human element. The design of a medical device must account for our psychology—our habits, assumptions, and cognitive biases.
The Lifecycle of a Medical Device
What happens after the 12 hours are up? The patch's journey isn't over. A used patch still contains a significant amount of active ingredient. Disposing of it carelessly—simply tossing it in an open bin—creates a serious risk.
A toddler or a pet doesn't see a medical device; they see something new to explore. Folding the patch in half, adhesive-side inward, and placing it securely out of reach is the final, critical step in its safe use. It's the closing clause in the contract.
A Quick Reference for Safe Application
To honor the complex engineering of this simple device, these key considerations are non-negotiable:
| Key Consideration | Action Required |
|---|---|
| Skin Condition | Apply only to intact, non-irritated skin. |
| Allergy & Sensitivity | Monitor for irritation; check for PABA derivative sensitivity. |
| External Factors | Avoid direct heat sources (heating pads, sunlight) over the patch. |
| Water Exposure | Do not swim, bathe, or shower during the 12-hour application. |
| Duration of Use | Adhere strictly to the 12-hour maximum wear time per 24 hours. |
| Safe Disposal | Fold adhesive sides together; discard safely away from children/pets. |
| Systemic Factors | Inform your doctor of liver conditions or other medications. |
Engineering Trust into Every Patch
The reliability of a transdermal patch—its consistent adhesion, predictable release rate, and chemical stability—is not an accident. It is the result of meticulous material science, rigorous quality control, and a deep understanding of the intricate system it operates within.
This is where the manufacturer's role becomes critical. For healthcare distributors and pharmaceutical brands, the goal is to provide patients with a product that honors this unseen contract every single time. It requires a partner who masters the complexities of transdermal R&D and bulk manufacturing. At Enokon, we specialize in producing reliable, FDA-compliant transdermal patches and pain plasters, ensuring that the simplicity your customers experience is backed by uncompromising science and engineering.
For healthcare brands and distributors seeking a partner who understands this intricate balance of chemistry, engineering, and human factors, let's build a solution together. Contact Our Experts
Visual Guide
Related Products
- Far Infrared Heat Pain Relief Patches Transdermal Patches
- Silicone Scar Sheets Patch Transdermal Drug Patch
- Icy Hot Menthol Medicine Pain Relief Patch
- Menthol Gel Pain Relief Patch
- Mugwort Wormwood Pain Relief Patch for Neck Pain
Related Articles
- Beyond the Pill: Why Transdermal Patches Are Redefining Pain Management
- Beyond the Pill: The Silent Engineering of Safer Pain Relief
- Beyond the Pill: The Engineering That Liberates Patients from Digestive Side Effects
- Beyond the Molecule: The Behavioral Science of the Simple Pain Patch
- The Silent Workhorse: How Transdermal Patches Rewrote the Rules of Pain Management




















