Comprehensive monitoring is non-negotiable when utilizing the methylphenidate transdermal system for treatment. Essential monitoring includes regular checks of cardiovascular health, specifically blood pressure and heart rate, both before and during therapy. For pediatric patients, diligent tracking of growth is critical, alongside careful observation for any adverse dermatological, psychiatric, or neurological effects.
Effective monitoring for transdermal methylphenidate is not a passive process. It is a systematic approach designed to proactively identify and manage potential cardiovascular, developmental, and dermatological risks to ensure patient safety and therapeutic efficacy.
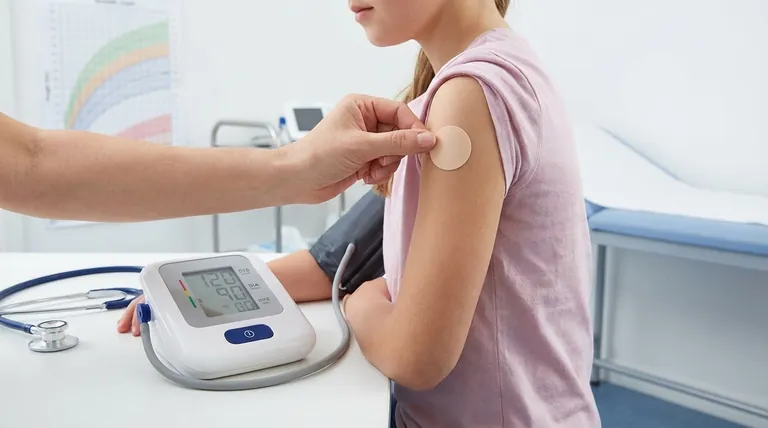
Foundational Monitoring: The Core Parameters
Consistent and regular check-ups are the bedrock of safe treatment. These visits allow for the assessment of both the medicine's effectiveness and the emergence of any unwanted effects.
Cardiovascular Assessment
Stimulant medications can place added stress on the cardiovascular system. Therefore, blood pressure and heart rate must be checked before initiating treatment to establish a baseline.
These vitals should be monitored regularly throughout the course of therapy to detect any significant increases that could pose a risk, particularly for individuals with pre-existing heart disease or defects.
Pediatric Growth and Development
The use of stimulants in children and adolescents requires close attention to physical development.
A patient's height and weight should be carefully monitored and tracked against growth charts. Any signs of growth suppression warrant a re-evaluation of the treatment plan.
General Efficacy and Side Effects
Regular appointments provide the opportunity to discuss the medicine's effectiveness and any subjective side effects.
This includes common issues like dizziness, drowsiness, or changes in vision that may affect daily activities such as driving or operating machinery.
Key Risks and Potential Complications to Monitor
Beyond the standard vitals, clinicians must remain vigilant for a range of specific, potentially serious complications associated with methylphenidate.
Dermatological Reactions at the Application Site
The transdermal delivery method introduces unique risks. The application site must be inspected for signs of contact sensitization, an allergic skin reaction.
Additionally, be aware of the potential for persistent loss of skin pigmentation (chemical leukoderma) at the application site, which can be permanent.
Psychiatric and Neurological Changes
This medication can impact mental and neurological status. It is crucial to monitor for new or worsening psychosis or other significant behavioral changes.
For patients with a history of seizures, be aware that methylphenidate may lower the convulsive threshold, requiring heightened vigilance.
Vascular and Circulatory Issues
Methylphenidate can affect the vascular system. Monitor for signs of peripheral vasculopathy, including Raynaud's phenomenon, which involves reduced blood flow to the extremities.
In male patients, it is important to be aware of the potential for priapism, a prolonged and painful erection that requires immediate medical attention.
Critical Patient Counseling Points
Effective monitoring involves empowering the patient with knowledge. Clear instructions on what to watch for and how to use the patch safely are essential components of the treatment plan.
Avoiding External Heat Sources
Patients must be explicitly warned against exposing the patch application site to direct heat, such as from heating pads, electric blankets, or prolonged sun exposure.
Heat can increase the rate of drug absorption, potentially leading to an overdose and a spike in side effects.
Managing Application and Dosing
Proper patch application is critical. Instruct patients to wear the patch for no longer than 9 hours and to follow a regular schedule.
Emphasize that they should never wear two patches at once to make up for a missed dose, as this significantly increases the risk of adverse effects.
A Practical Monitoring Framework
Tailor your monitoring strategy based on the specific patient profile and treatment goals.
- If the patient is a child or adolescent: Prioritize regular monitoring of height, weight, blood pressure, and heart rate at every visit.
- If the patient has any cardiac risk factors: Implement baseline and frequent follow-up cardiovascular assessments before and during treatment.
- If the patient reports any skin irritation: Immediately inspect the application site for signs of contact sensitization or pigment changes.
- If there is a pre-existing psychiatric or seizure history: Maintain heightened vigilance for any changes in mood, behavior, or neurological symptoms.
A proactive and systematic monitoring plan is the cornerstone of safe and effective therapy with transdermal methylphenidate.
Summary Table:
| Monitoring Area | Key Parameters to Track | Frequency / Notes |
|---|---|---|
| Cardiovascular | Blood Pressure, Heart Rate | Baseline & regularly during therapy |
| Pediatric Growth | Height, Weight | Track against growth charts at each visit |
| Dermatological | Skin reaction at application site (allergy, pigmentation loss) | Inspect site regularly |
| Psychiatric/Neurological | New/worsening psychosis, behavioral changes, seizures | Maintain vigilance, especially with history |
| Vascular | Signs of peripheral vasculopathy (e.g., Raynaud's), Priapism | Monitor for symptoms |
Ensure the highest safety standards for your transdermal products. Partner with Enokon, a bulk manufacturer of reliable transdermal patches and pain plasters for healthcare and pharma distributors and brands. Benefit from our technical expertise for custom R&D and development, ensuring your products are designed with patient safety and monitoring needs in mind. Contact our experts today to discuss your transdermal project.
Visual Guide
Related Products
- Menthol Gel Pain Relief Patch
- Far Infrared Heat Pain Relief Patches Transdermal Patches
- Icy Hot Menthol Medicine Pain Relief Patch
- Silicone Scar Sheets Patch Transdermal Drug Patch
- Mugwort Wormwood Pain Relief Patch for Neck Pain
People Also Ask
- How effective are pain relief patches for muscle pain? Target Localized Pain with Transdermal Delivery
- Who should consult a healthcare professional before using pain relief patches? Ensure Your Safety with Medical Advice
- What are the potential side effects of pain relief patches? A Guide to Safe Use & Key Risks
- What are some common applications of pain relief patches? Effective Relief for Muscle & Joint Pain
- How do pain relief patches compare to oral painkillers? Targeted Relief vs. Systemic Effects



















