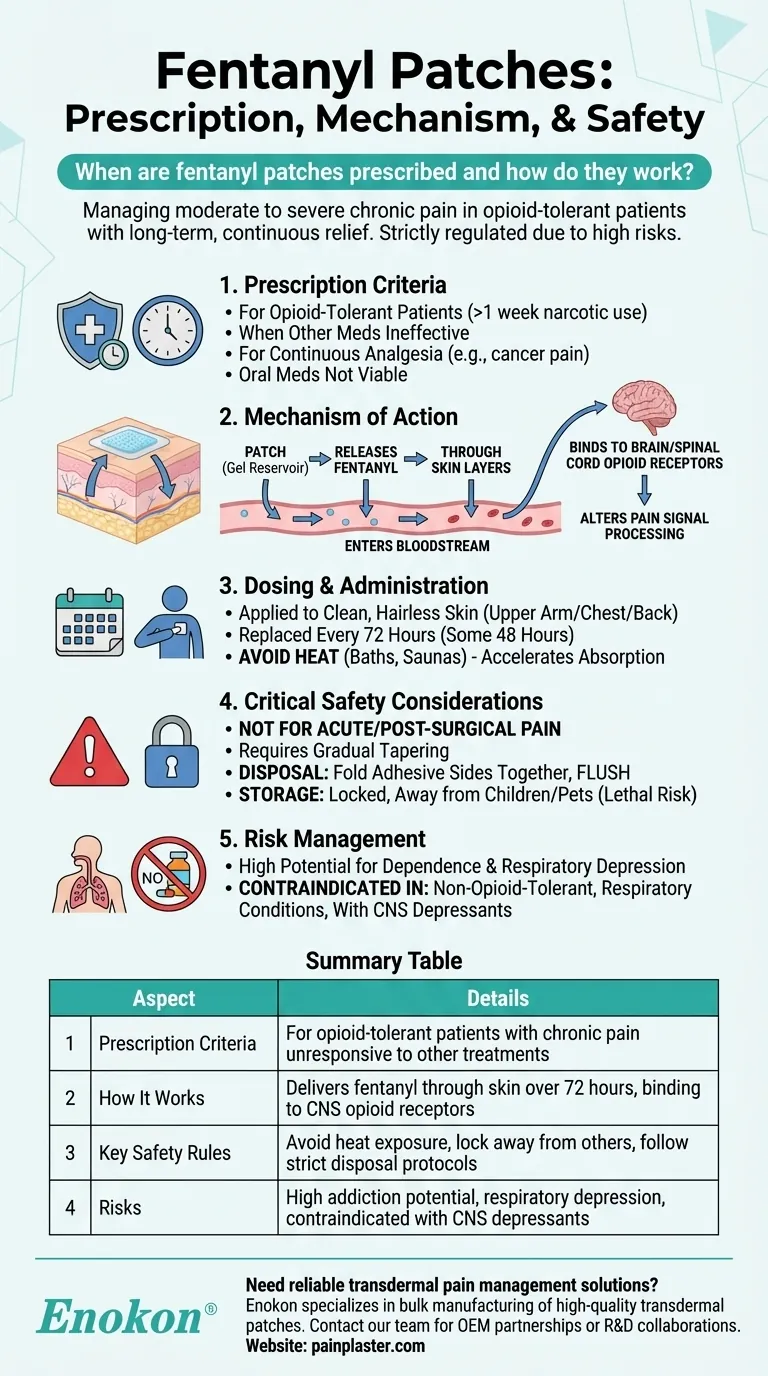
Fentanyl patches are prescribed for managing moderate to severe chronic pain in opioid-tolerant patients who require long-term, continuous pain relief. These transdermal patches deliver the potent opioid fentanyl through the skin over 72 hours, altering pain perception in the nervous system. Their use is strictly regulated due to high addiction potential and overdose risks, requiring careful patient selection and disposal protocols.
Key Points Explained:
-
Prescription Criteria
- Reserved for opioid-tolerant patients (those taking regular narcotic medication for >1 week)
- Used when:
- Other pain medications prove ineffective
- Continuous analgesia is needed for chronic conditions (e.g., cancer pain)
- Oral medications aren't viable options
-
Mechanism of Action
- The Fentanyl Patch contains a gel reservoir that:
- Slowly releases fentanyl through skin layers
- Enters bloodstream for systemic distribution
- Binds to brain/spinal cord opioid receptors
- Alters pain signal processing by inhibiting neurotransmitter release
- The Fentanyl Patch contains a gel reservoir that:
-
Dosing & Administration
- Applied to clean, hairless skin (upper arm/chest/back)
- Replaced every 72 hours (some variants last 48 hours)
- Heat exposure accelerates absorption (avoid hot baths/saunas)
-
Critical Safety Considerations
- Not for acute/post-surgical pain due to overdose risk
- Requires gradual tapering to prevent withdrawal
- Disposal protocol: Fold adhesive sides together before flushing
- Storage: Locked away from children/pets (lethal to non-tolerant individuals)
-
Risk Management
- High potential for dependence and respiratory depression
- Contraindicated in:
- Non-opioid-tolerant patients
- Those with respiratory conditions
- Patients taking CNS depressants (e.g., benzodiazepines)
These patches represent a balance between effective chronic pain management and substantial public health risks, demanding vigilant clinical oversight. Their transdermal technology exemplifies how targeted drug delivery systems can improve quality of life for carefully selected patients.
Summary Table:
| Aspect | Details |
|---|---|
| Prescription Criteria | For opioid-tolerant patients with chronic pain unresponsive to other treatments |
| How It Works | Delivers fentanyl through skin over 72 hours, binding to CNS opioid receptors |
| Key Safety Rules | Avoid heat exposure, lock away from others, follow strict disposal protocols |
| Risks | High addiction potential, respiratory depression, contraindicated with CNS depressants |
Need reliable transdermal pain management solutions?
Enokon specializes in bulk manufacturing of high-quality transdermal patches, including custom formulations for chronic pain relief. Our technical expertise ensures precise drug delivery systems tailored to your requirements.
Contact our team to discuss OEM partnerships or R&D collaborations for your pain management product line.
Visual Guide
Related Products
- Menthol Gel Pain Relief Patch
- Icy Hot Menthol Medicine Pain Relief Patch
- Lidocaine Hydrogel Pain Relief Patch for Pain Relief
- Far Infrared Pain Patch Relief Pain Reliever for Back
- Far Infrared Heat Pain Relief Patches Transdermal Patches
People Also Ask
- How effective are pain relief patches for muscle pain? Target Localized Pain with Transdermal Delivery
- What are pain relief patches and how do they work? Discover Non-Invasive Pain Management
- Who should consult a healthcare professional before using pain relief patches? Ensure Your Safety with Medical Advice
- Can pregnant women use pain relief patches? Your Essential Guide to Safe Pain Management
- What are the potential side effects of pain relief patches? A Guide to Safe Use & Key Risks



















