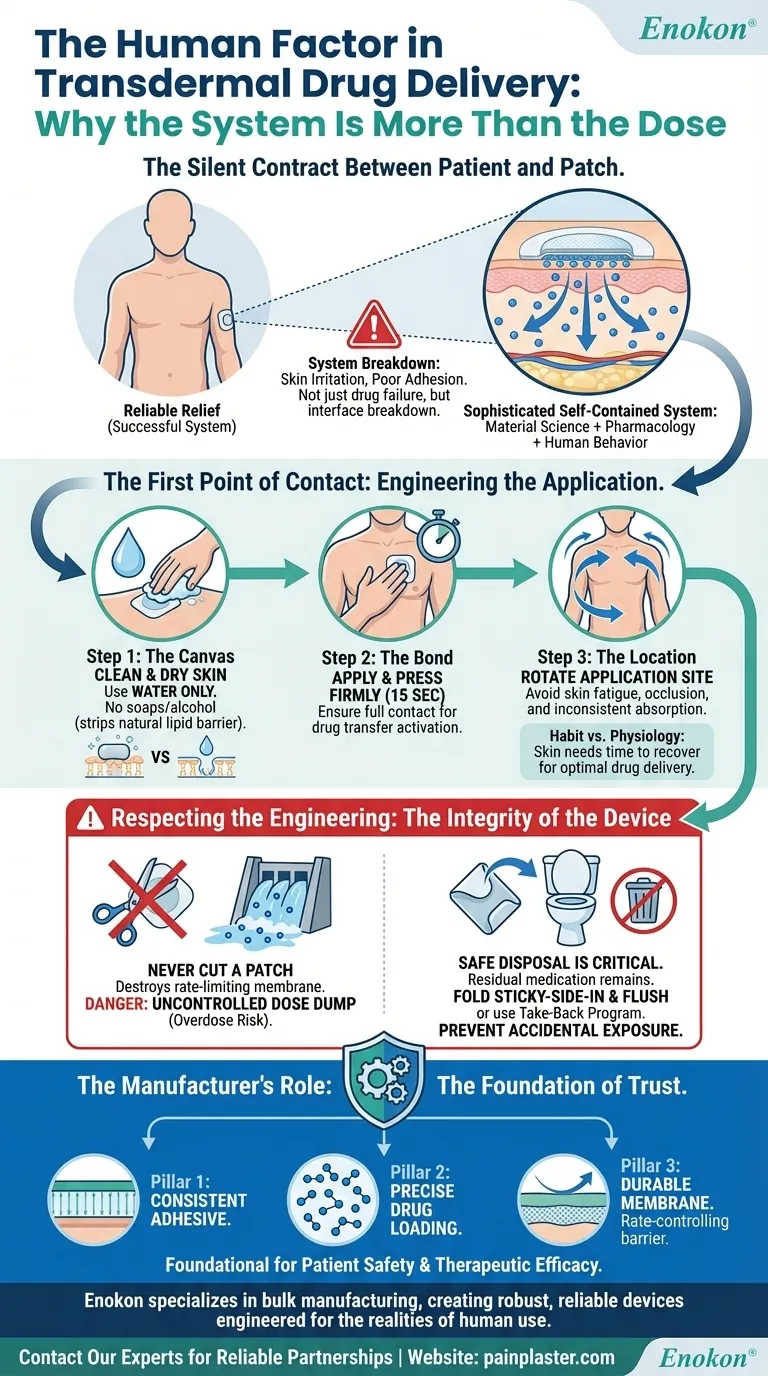
The Silent Contract Between Patient and Patch
Imagine a patient managing chronic pain. A small transdermal patch, applied once a week, provides a steady, reliable stream of relief. For months, it's a marvel of modern medicine. Then, a subtle shift occurs. The skin beneath the patch becomes irritated. Adhesion seems less certain.
This isn't a failure of the drug. It's a breakdown in the system.
A transdermal patch is not merely a sticker with medicine inside. It is a sophisticated, self-contained drug delivery system. Its success relies on a delicate interface between material science, pharmacology, and the most unpredictable variable of all: human behavior.
The First Point of Contact: Engineering the Application
The seven-day therapeutic journey of a buprenorphine patch begins with a few critical moments of application. These instructions are not arbitrary suggestions; they are the parameters required to initialize the system correctly.
- The Canvas: Skin must be clean, dry, and intact. Why wash with water only, not soap? Soaps and alcohols can strip the skin's natural lipid barrier, altering the very surface the patch's adhesive and permeation enhancers are engineered to work with.
- The Bond: The patch must be applied to an area with minimal hair and flexion—the upper arm, chest, or back. A firm 15-second press isn't about just making it stick; it ensures full contact between the adhesive matrix and the epidermis, activating the thermal and physical properties that initiate drug transfer.
- The Location: Application sites must be rotated. This is perhaps the most critical and most frequently overlooked behavioral requirement.
The Psychology of Habit vs. The Physiology of Skin
Humans are creatures of habit. It is cognitively easier to apply the patch to the same familiar, accessible spot each week. But the skin is a living organ.
Constant application to a single site leads to localized skin fatigue. The stratum corneum can become occluded, irritated, or saturated. This not only causes discomfort but can subtly compromise the consistent rate of drug absorption the patch was designed to deliver.
Rotating the application site isn't just about avoiding a rash. It is a deliberate act of system maintenance, allowing the skin to fully recover and ensuring each new patch is applied to a fresh, optimally receptive surface.
Respecting the Engineering: The Integrity of the Device
The power of a buprenorphine patch lies in its controlled-release mechanism. A complex series of layers and membranes precisely governs the rate at which the potent opioid crosses the skin barrier. This is why the physical integrity of the patch is absolute.
- Never Cut a Patch: Cutting or damaging a patch destroys its rate-limiting membrane. This is akin to breaking a dam. The result is an uncontrolled, unpredictable release of the drug—a "dose dump" that can lead to a dangerous overdose. The device must be used exactly as it was engineered.
- The End of the Lifecycle: Even after seven days, a used patch contains a significant amount of residual medication. Disposal is the final, critical safety protocol. Simply tossing it in the trash creates a risk of accidental exposure to children, pets, or others. Folding it sticky-side-in and flushing or using a take-back program deactivates the system safely.
The Manufacturer's Role: The Foundation of Trust
The reliability of this entire human-device system begins long before a patient ever opens the box. It begins with the manufacturer.
The consistency of the adhesive, the precise loading of the active pharmaceutical ingredient, and the durability of the rate-controlling membrane are not just line items on a quality-control checklist. They are the foundational pillars upon which patient safety and therapeutic efficacy are built. A patch that fails to adhere for the full seven days, or one with inconsistent drug delivery, undermines the entire system, regardless of how well the patient follows instructions.
This is where a deep understanding of transdermal technology is non-negotiable. At Enokon, we specialize in the bulk manufacturing of transdermal patches and pain plasters, recognizing that we are not just producing a product, but a critical component of a patient's therapeutic system. Our R&D and manufacturing processes are built around creating devices that are robust, reliable, and engineered for the realities of human use.
For healthcare distributors and pharmaceutical brands seeking a partner who grasps this deep integration of engineering and human factors, the conversation starts with a reliable foundation. Contact Our Experts
Visual Guide
Related Products
- Silicone Scar Sheets Patch Transdermal Drug Patch
- Cooling Fever Patches Color Change Cold Fever Patch
- Far Infrared Heat Pain Relief Patches Transdermal Patches
- Icy Hot Menthol Medicine Pain Relief Patch
- Menthol Gel Pain Relief Patch
Related Articles
- The Paradox of the Patch: Deconstructing the Science of Skin-Contact Safety
- Beyond the Pill: The Silent Engineering of Safer Pain Relief
- Beyond the Counter: The Hidden Engineering of Trust in Pain Management
- The Interface Problem: Engineering Transdermal Patches That Respect the Skin
- The Unseen Chemistry: Why Transdermal Patch Efficacy Fades on the Shelf