The Final, Forgotten Mile of Drug Delivery
The most sophisticated systems often fail at their simplest points. A transdermal patch—a small, unassuming square applied to the skin—is a marvel of pharmaceutical engineering, designed for steady, controlled drug delivery. Yet its journey doesn't end when it's peeled off.
The final mile of its lifecycle, from the moment it leaves the package to its ultimate disposal, is fraught with invisible risks. A patch left in a hot car, a used one tossed carelessly into an open bin—these small oversights can compromise patient safety, negate therapeutic effects, and harm the environment. This isn't just about user error; it's about understanding the patch as the precision system it truly is.
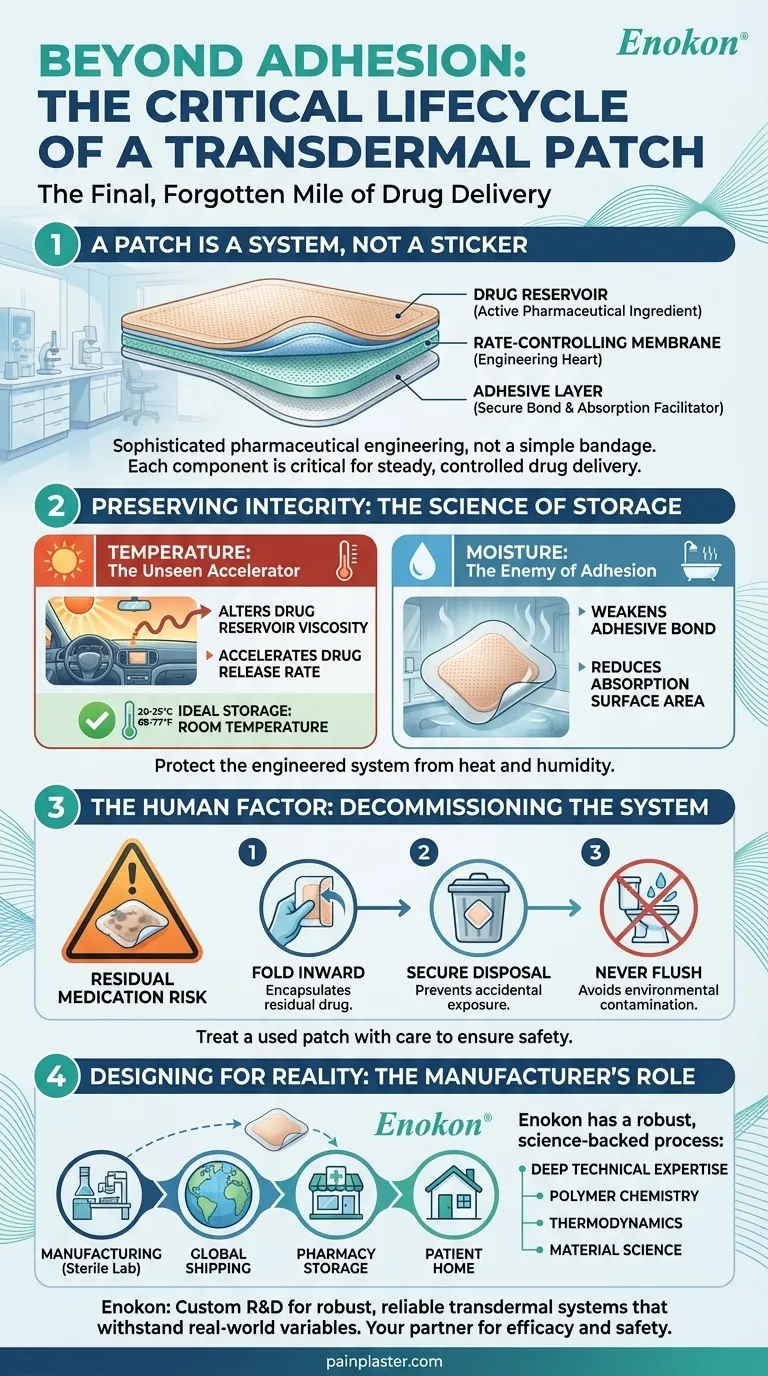
A Patch is a System, Not a Sticker
From a psychological standpoint, we tend to underestimate the familiar. Because a patch looks and feels like a simple bandage, we treat it as such. This mental shortcut is dangerous.
A transdermal patch is a multi-layered drug delivery system, where each component is critical:
- The Drug Reservoir: Contains the active pharmaceutical ingredient (API), like lidocaine, in a precise concentration.
- The Rate-Controlling Membrane: The engineering heart of the patch, ensuring the drug is released at a consistent, predictable rate over hours.
- The Adhesive Layer: More than just glue, this layer must secure the system to the skin without causing irritation, while also facilitating drug absorption.
Any disruption to this system compromises its function. The strict handling protocols aren't arbitrary rules; they are the operating manual for maintaining the system's integrity.
Preserving Integrity: The Science of Storage
Proper storage is the first line of defense in protecting the engineered system. The primary enemies are heat and moisture, each attacking the patch in a different way.
Temperature: The Unseen Accelerator
Storing a patch at room temperature (20-25°C / 68-77°F) is non-negotiable. Exposing it to heat—whether from a sunlit dashboard, a heating pad, or improper warehouse storage—does more than just degrade the ingredients.
Heat can alter the viscosity of the drug reservoir and the permeability of the membrane. This accelerates the drug release rate, transforming a steady, 12-hour dose into an unpredictable, potentially excessive one. The patch's fundamental promise of controlled release is broken.
Moisture: The Enemy of Adhesion
A bathroom cabinet, with its fluctuating humidity, is one of the worst places to store transdermal patches. Moisture attacks the adhesive layer, weakening its bond.
If a patch’s edges lift or it fails to adhere properly, the surface area for drug absorption is reduced, leading to under-dosing. The therapeutic benefit is lost, not because the drug is flawed, but because the delivery system was compromised before it was ever used.
The Human Factor: Decommissioning the System
A used patch is not inert. A significant amount of the active drug can remain even after the recommended wear time. This residual medication poses a serious risk, especially to children and pets who might accidentally touch or ingest a discarded patch.
A Simple Protocol for Safety
The disposal process is, in effect, the system's decommissioning protocol.
| Action | Rationale |
|---|---|
| Fold Inward | Fold the patch in half, adhesive sides together. This encapsulates the residual drug, making it inaccessible. |
| Secure Disposal | Place it in a lidded trash can. This prevents accidental exposure to others. |
| Never Flush | Flushing introduces APIs into water systems, harming aquatic life and polluting the environment. |
Treating a used patch with this level of care acknowledges its lingering potency. It’s the final step in responsible medication management.
Designing for Reality: The Manufacturer's Role
For pharmaceutical brands and distributors, the choice of a manufacturing partner is a choice about system reliability. A manufacturer’s job is not just to create a patch that works in a sterile lab, but one that remains stable and safe through the chaos of global shipping, pharmacy storage, and real-world patient homes.
This is where deep technical expertise becomes paramount. A partner must understand the polymer chemistry of adhesives, the thermodynamics of drug release, and the material science of protective packaging. They must design for the realities of a patch left in a travel bag or stored in a humid climate.
As a bulk manufacturer, Enokon is built on this principle. Our custom R&D and development processes are focused on creating robust and reliable transdermal systems that account for these real-world variables. We help our clients deliver not just a product, but a complete therapeutic system designed for efficacy and safety throughout its entire lifecycle. For distributors and brands dedicated to this level of quality, partnering with a technical expert is the first step. Contact Our Experts
Visual Guide
Related Products
- Far Infrared Heat Pain Relief Patches Transdermal Patches
- Silicone Scar Sheets Patch Transdermal Drug Patch
- Icy Hot Menthol Medicine Pain Relief Patch
- Menthol Gel Pain Relief Patch
- Mugwort Wormwood Pain Relief Patch for Neck Pain
Related Articles
- The Silent Workhorse: How Transdermal Patches Rewrote the Rules of Pain Management
- Beyond the Pill: The Physics and Psychology of Transdermal Pain Relief
- Beyond Efficacy: The Psychology of Adherence in Topical Pain Relief
- Engineering Certainty: The Invisible Architecture of a Gold-Standard Clinical Trial
- The Human Interface of Pain Relief: Why Delivery Systems Define Success




















