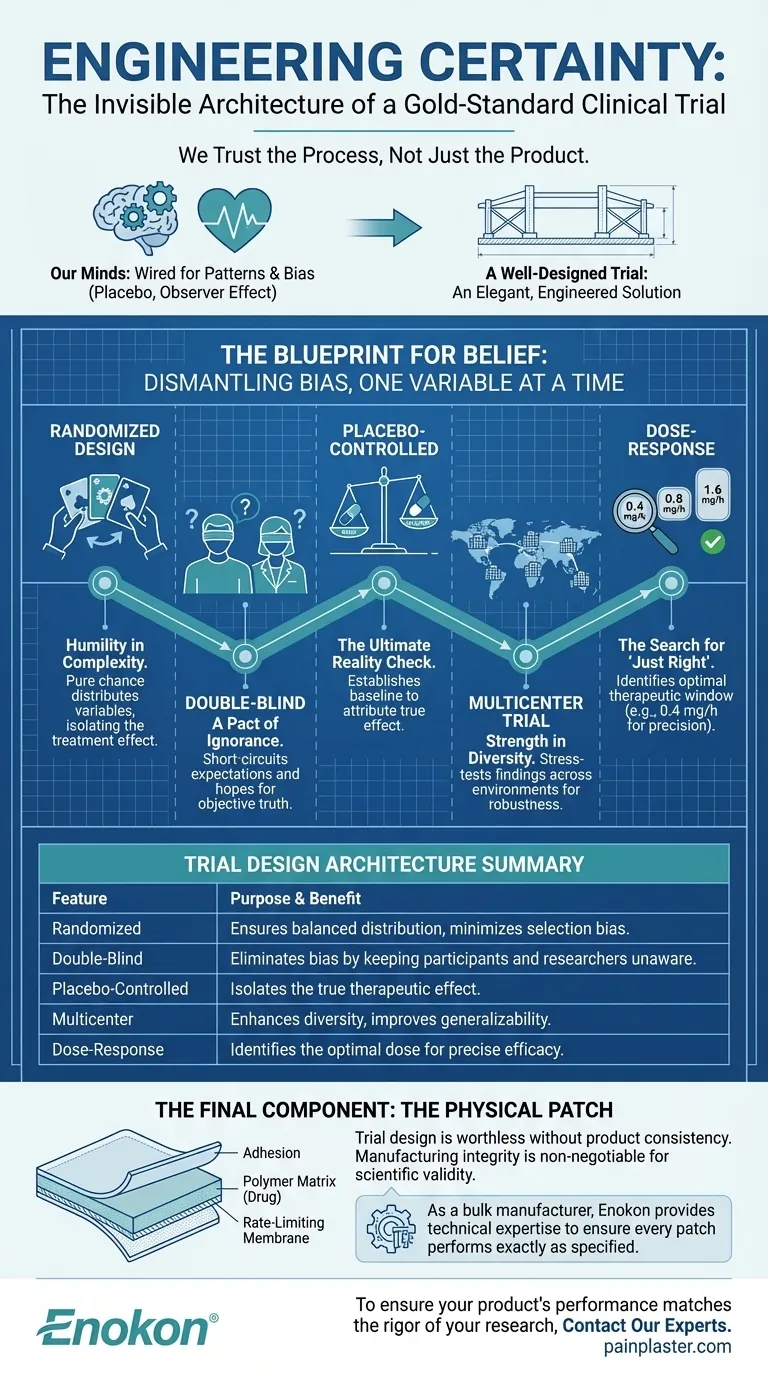
We Trust the Process, Not Just the Product
Why do we trust a medicine?
The answer seems obvious: because it works. But the real story is deeper. We trust a medicine because of the invisible architecture of the process that validated it. We trust the blueprint that dismantled doubt and systematically eliminated bias.
Our minds are wired to find patterns and take cognitive shortcuts. This is a feature, not a bug, for everyday life. But in science, it’s a liability. The placebo effect, where belief alone can generate a physiological response, is a powerful testament to this. So is observer bias, where researchers unconsciously see the results they hope for.
A well-designed clinical trial is an elegant, engineered solution to these deeply human problems. Let's deconstruct the "gold standard" design using a real-world study on a nitroglycerin transdermal patch for heart attack survivors.
The Blueprint for Belief
A clinical trial isn't just an experiment; it's a carefully constructed system designed to isolate one variable: the effect of the drug itself. The nitroglycerin patch study used a randomized, double-blind, placebo-controlled, multicenter design.
Each of those terms represents a pillar in the architecture of certainty.
Dismantling Bias, One Variable at a Time
To get to the truth, you must first create an environment where the truth can be seen clearly, free from the noise of human psychology.
-
Randomized Design: Humility in the Face of Complexity Participants were randomly assigned to receive either a placebo or one of three active patch doses. Randomization is an act of scientific humility. It accepts that countless known and unknown variables (age, genetics, lifestyle) could influence the outcome. Rather than trying to control them all—an impossible task—it lets pure chance distribute them evenly across all groups. It ensures the only systematic difference between groups is the treatment itself.
-
Double-Blind: A Pact of Ignorance for Objective Truth In this study, neither the patients nor the researchers knew who was getting the real patch and who was getting the placebo. This is the double-blind. It short-circuits our biases. The patient's expectation can't create a placebo effect, and the researcher's hope can't color their interpretation of the data. It's a temporary, shared ignorance in the service of a permanent, objective truth.
-
Placebo-Controlled: The Ultimate Reality Check Including a placebo group answers the most fundamental question: Is this treatment better than doing nothing? The placebo provides a baseline. Any effect observed in the active treatment groups beyond what's seen in the placebo group can be confidently attributed to the drug itself.
From a Single Lab to the Real World
Isolating the effect is only half the battle. The findings must also be reliable and applicable to the broad patient population who will eventually use the treatment.
-
Multicenter Trial: Strength in Diversity The study was conducted across multiple hospitals. A result found in a single location might be a fluke, influenced by local demographics or clinical practices. A multicenter trial stress-tests the findings across different environments, ensuring the results are robust and generalizable.
-
Dose-Response: The Search for "Just Right" The trial didn't just ask "does it work?" but "at what dose does it work best?" By testing three different doses (0.4, 0.8, and 1.6 mg/h), researchers identified a specific therapeutic window. Interestingly, only the lowest 0.4-mg/h dose showed a statistically significant benefit. This isn't a failure; it's precision. It prevents patients from being over-medicated and defines the exact target for optimal effect.
This table summarizes the architecture of the trial:
| Trial Design Feature | Purpose & Benefit |
|---|---|
| Randomized | Ensures balanced group distribution and minimizes selection bias. |
| Double-Blind | Eliminates bias by keeping participants and researchers unaware of groups. |
| Placebo-Controlled | Isolates the true therapeutic effect of the nitroglycerin patch. |
| Multicenter | Enhances participant diversity and improves generalizability of results. |
| Dose-Response Testing | Identifies the optimal dose (0.4 mg/h showed significant benefits). |
| Targeted Population | Focused on AMI survivors for precise efficacy evaluation. |
The Final Component: The Physical Patch
Here’s the thing. The most elegant trial design in the world is worthless if the physical product being tested is inconsistent.
Imagine if the "0.4 mg/h" patches in the study were poorly manufactured. Some might release 0.3 mg/h, others 0.5 mg/h. This variability would introduce chaos into the meticulously controlled environment of the trial, muddying the data and potentially obscuring a real therapeutic effect.
Manufacturing consistency is not just a quality control issue; it is a prerequisite for scientific validity. The transdermal patch itself—its adhesion, its polymer matrix, its rate-limiting membrane—must be as reliable as the trial methodology. The engineering of the patch is as critical as the engineering of the study.
This is where the blueprint of the trial meets the reality of production. For pharmaceutical brands and distributors running these critical studies or bringing a validated product to market, the physical integrity of the delivery system is non-negotiable. As a bulk manufacturer specializing in transdermal patches, Enokon provides the technical expertise in R&D and manufacturing to ensure every single patch performs exactly as specified. We build the physical certainty that your clinical science relies upon.
To ensure your product's performance matches the rigor of your research, Contact Our Experts.
Visual Guide
Related Products
- Far Infrared Heat Pain Relief Patches Transdermal Patches
- Silicone Scar Sheets Patch Transdermal Drug Patch
- Icy Hot Menthol Medicine Pain Relief Patch
- Menthol Gel Pain Relief Patch
- Mugwort Wormwood Pain Relief Patch for Neck Pain
Related Articles
- Beyond the Pill: The Silent Engineering of Safer Pain Relief
- The Human Interface of Pain Relief: Why Delivery Systems Define Success
- Beyond Removal: The Hidden Pharmacokinetics of Transdermal Patches
- Beyond the Pill: Why Transdermal Patches Are Redefining Pain Management
- Beyond the Molecule: The Behavioral Science of the Simple Pain Patch




















