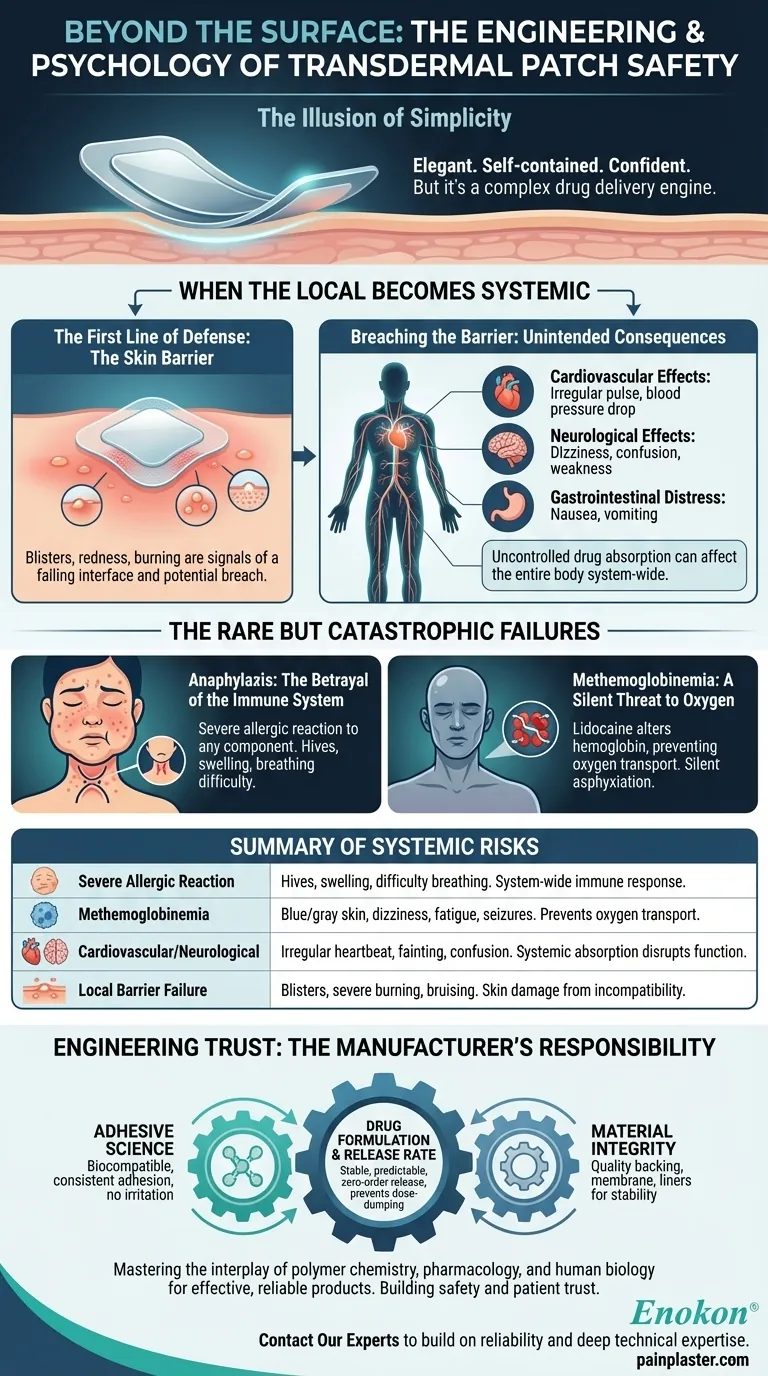
The Illusion of Simplicity
A transdermal patch is an elegant solution. Clean, self-contained, and applied directly to the site of pain, it represents the peak of user-friendly medicine. Its very design inspires confidence.
This perception of simplicity is a powerful psychological advantage. We trust things we understand, and a patch feels more like a bandage than a complex pharmacological system. But this is where the danger lies. A transdermal patch is not a passive covering; it's an active, sophisticated drug delivery engine.
When that engine malfunctions—due to poor design, improper use, or unforeseen biological reactions—the consequences can move from localized to systemic, sometimes with alarming speed.
When the Local Becomes Systemic
The contract between a patch and a patient is that the effect will stay where it's put. The first sign of a broken contract often appears right on the skin.
The First Line of Defense: The Skin Barrier
Blisters, severe redness, bruising, or a burning sensation at the application site are more than minor annoyances. They are signals that the first interface—the delicate balance between the patch's adhesive, the drug concentration, and the patient's skin—is failing. This isn't just an irritation; it's a potential breach in the body's primary defense.
Breaching the Barrier: Unintended Consequences
Once the drug's delivery is no longer perfectly controlled, it can be absorbed into the bloodstream at unintended rates. Lidocaine, designed to block local nerve signals, can begin affecting the entire body.
The symptoms of this systemic breach can include:
- Cardiovascular Effects: An irregular or rapid pulse, a drop in blood pressure, or fainting. Lidocaine is used medically to control heart rhythms, so an uncontrolled dose can have potent effects on the heart.
- Neurological Effects: Dizziness, confusion, blurred vision, or weakness. The same nerve-blocking action that soothes a sore muscle can disrupt the central nervous system.
- Gastrointestinal Distress: Nausea and vomiting are common signs that a substance has entered the bloodstream and is affecting the body system-wide.
The Rare but Catastrophic Failures
Most systems have failure modes that are rare but severe. In transdermal patches, these represent a complete breakdown of the intended therapeutic action, turning a source of relief into a medical emergency.
Anaphylaxis: The Betrayal of the Immune System
A severe allergic reaction is a catastrophic event. Hives, swelling of the face and throat, and difficulty breathing can be triggered not just by the active ingredient, but by any component in the patch's adhesive or matrix. It is a stark reminder that every material touching the body must be chosen with immense care for biocompatibility.
Methemoglobinemia: A Silent Threat to Oxygen
Perhaps the most insidious risk is methemoglobinemia. In this rare condition, the lidocaine absorbed into the blood alters hemoglobin, the molecule that carries oxygen. The blood can no longer deliver oxygen effectively to the tissues.
The result is a silent, creeping asphyxiation, with symptoms like blue-gray skin, profound fatigue, and seizures. It's a prime example of how a seemingly simple local therapy can cause a complex and life-threatening internal crisis.
Summary of Systemic Risks
| Risk Category | Key Symptoms | Underlying Mechanism |
|---|---|---|
| Severe Allergic Reaction | Hives, swelling, difficulty breathing | System-wide immune response (anaphylaxis) to a patch component. |
| Methemoglobinemia | Blue/gray skin, dizziness, fatigue, seizures | Lidocaine alters hemoglobin, preventing oxygen transport. |
| Cardiovascular/Neurological | Irregular heartbeat, fainting, confusion | Systemic absorption disrupts heart and central nervous system function. |
| Local Barrier Failure | Blisters, severe burning, bruising | Chemical or adhesive incompatibility leading to skin damage. |
Engineering Trust: The Manufacturer's Responsibility
These risks are not inevitable. They are engineering problems that can be solved with expertise and precision. The safety of a transdermal patch is determined long before it reaches the patient, in the design and manufacturing process.
A manufacturer's control over these variables is absolute:
- Adhesive Science: Selecting a biocompatible adhesive that provides consistent adhesion without causing skin irritation is the first and most critical step.
- Drug Formulation & Release Rate: The core of the system is the drug matrix. Engineering it for a stable, predictable, zero-order release kinetic is crucial to prevent dose-dumping and systemic absorption.
- Material Integrity: The quality of the backing, membrane, and liners ensures the patch remains stable and its contents secure until the moment of application.
For manufacturers like Enokon, this is the central challenge. It involves mastering the interplay between polymer chemistry, pharmacology, and human biology to create a product that is not only effective but fundamentally reliable. This expertise allows brands and distributors to build product lines founded on safety and patient trust.
Building the next generation of transdermal therapies requires a partner who understands that a simple patch is a promise of safety. If you're looking to develop a product line built on reliability and deep technical expertise, Contact Our Experts.
Visual Guide
Related Products
- Far Infrared Heat Pain Relief Patches Transdermal Patches
- Silicone Scar Sheets Patch Transdermal Drug Patch
- Icy Hot Menthol Medicine Pain Relief Patch
- Menthol Gel Pain Relief Patch
- Mugwort Wormwood Pain Relief Patch for Neck Pain
Related Articles
- Beyond Efficacy: The Psychology of Adherence in Topical Pain Relief
- Beyond the Molecule: The Behavioral Science of the Simple Pain Patch
- The Silent Workhorse: How Transdermal Patches Rewrote the Rules of Pain Management
- Engineering Certainty: The Invisible Architecture of a Gold-Standard Clinical Trial
- Beyond Removal: The Hidden Pharmacokinetics of Transdermal Patches